AI Regulatory Compliance Document Processing Agent

Automating Regulatory Compliance Document Processing with AI
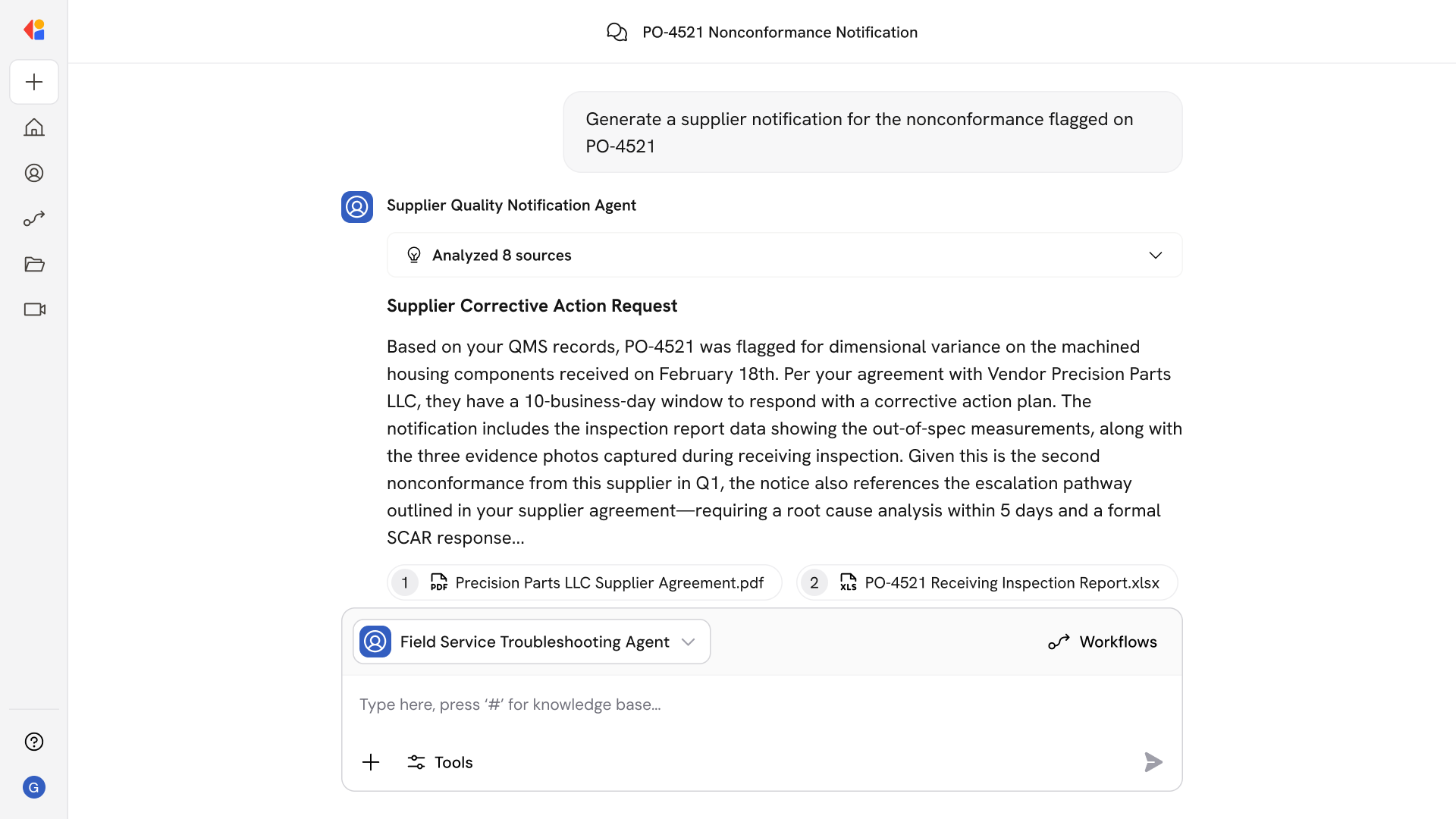
Obligation Extraction and Structuring
The agent uses NLP to identify requirements, conditions, thresholds, and deadlines from regulatory text—building a structured obligation register with full citation lineage.
Requirements-to-Controls Mapping
Automated semantic matching links extracted obligations to your existing control library, SOPs, and work instructions, maintaining a living traceability matrix that highlights gaps requiring remediation.
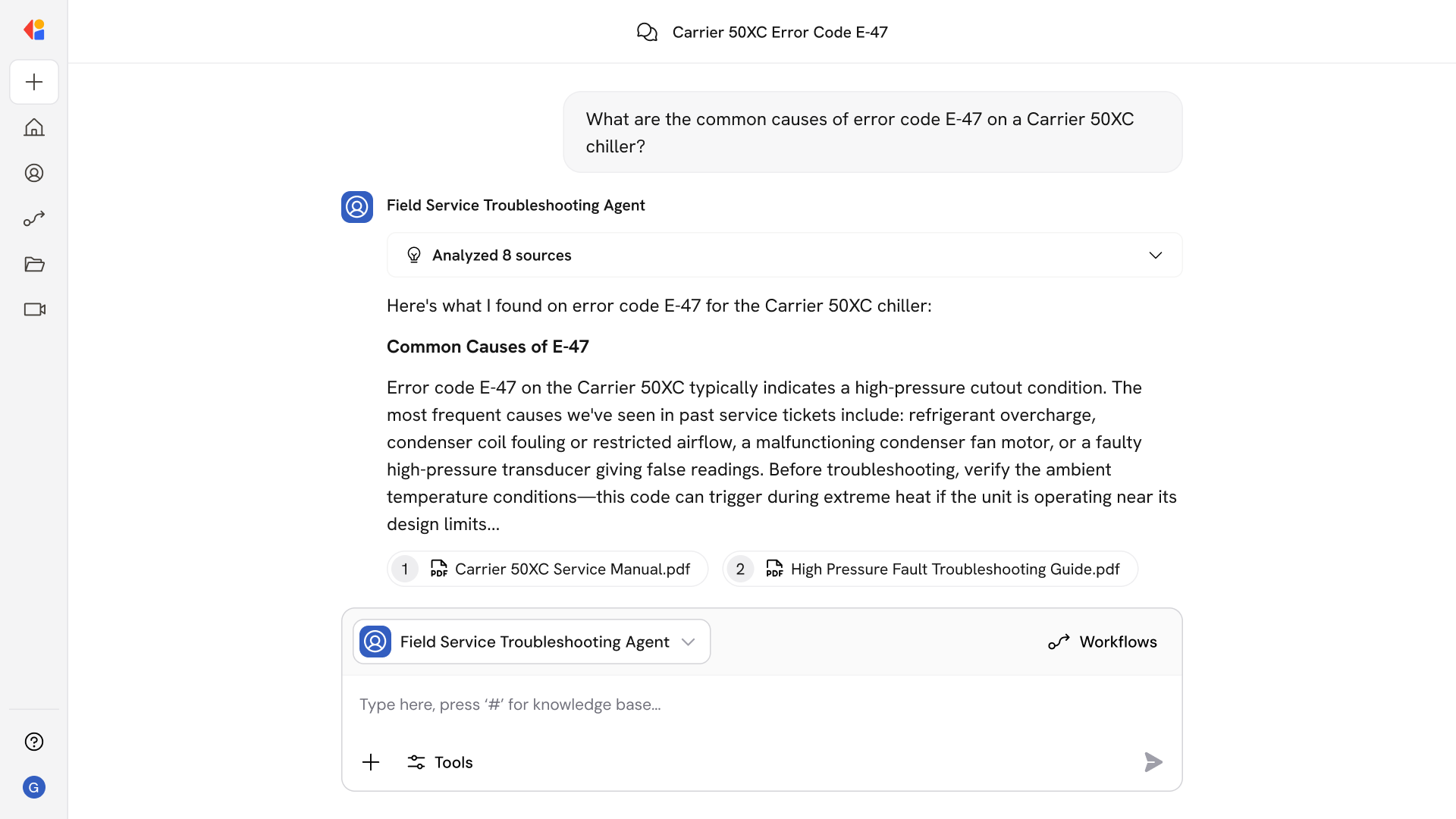
Audit-Ready Evidence Collection
The system harvests timestamped artifacts from connected business systems—batch records, calibration certificates, training completions—and compiles them into defensible audit binders with full provenance.
How Cassidy automates regulatory compliance using AI
Step 1: Ingest regulatory documents
The Workflow triggers when new regulations, standards, or guidance documents are added—whether from FDA, ISO, EU MDR, or internal uploads. Cassidy normalizes the content and tags it with jurisdiction, applicability, and version metadata.
Step 2: Extract and structure obligations
Cassidy's Knowledge Base and AI Agents parse the regulatory text to identify specific obligations, conditions, timelines, and citations. Each requirement is added to a centralized obligation register with owner assignments and due dates.
Step 3: Map to controls and SOPs
The Workflow matches extracted obligations against your existing control library and SOPs using semantic similarity. Cassidy flags gaps where no control exists and surfaces conflicts or overlaps requiring SME review.
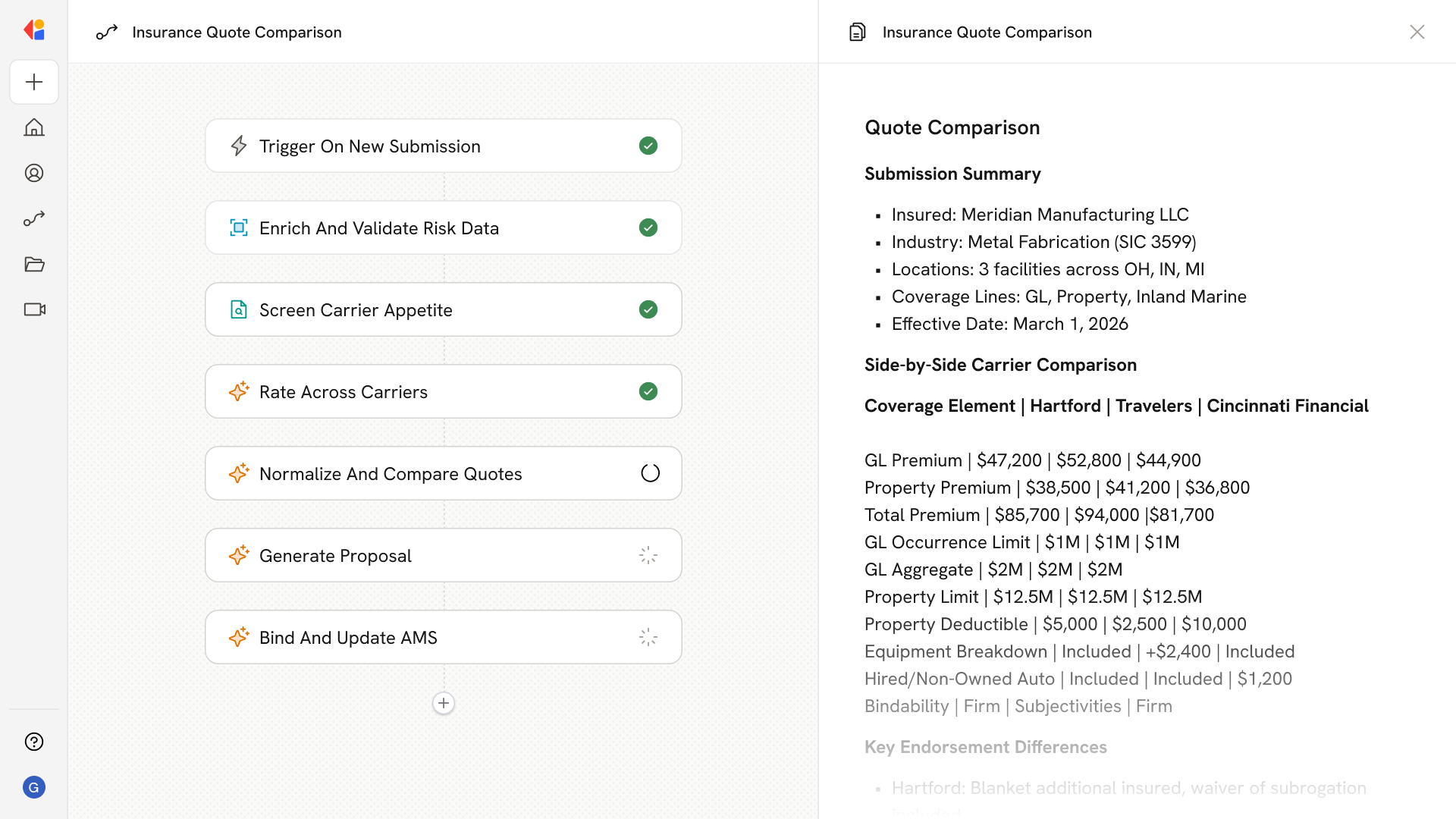
Step 4: Generate gap analysis and remediation tasks
Cassidy produces a prioritized gap report with risk scoring and automatically creates change control tasks in your connected QMS or project management system, assigning owners and deadlines.
Step 5: Draft SOP updates
For identified gaps, Cassidy generates redlined SOP language aligned to specific regulatory clauses—ready for SME review, approval workflows, and version-controlled document management.
Step 6: Harvest and link evidence
Cassidy integrates with your ERP, MES, LIMS, and QMS to pull timestamped evidence artifacts. Each piece of evidence is linked to specific controls and time periods, maintaining ALCOA+ data integrity.
Step 7: Compile audit-ready binders
On demand or on schedule, Cassidy assembles complete audit packages—traceability matrices, control narratives, validation documentation, and linked evidence—with full provenance and approval history.
Implement it inside your company
- Hands-on onboarding and support
- Self-paced training for your team
- Dedicated implementation experts
- Ongoing use case discovery
- ROI tracking & analytics dashboards
- Proven playbooks to get started fast