AI Quality Improvement Knowledge Agent
Automating Healthcare Quality Improvement with AI
Grounded answers with auditable provenance
Every response draws from versioned clinical guidelines, measure specifications, and local policies—with full citation trails that satisfy regulatory scrutiny and governance requirements.
Intelligent measure computation and SPC charting
The agent pulls data from EHR and claims systems, calculates numerators and denominators with proper exclusions, and generates annotated run and control charts that reveal special-cause variation and centerline shifts.
Living knowledge that evolves with your organization
KCS-driven workflows capture frontline insights, link cases to knowledge articles, and continuously refine content—eliminating stale policies and scattered documentation across committees and intranets.
How Cassidy automates this using AI
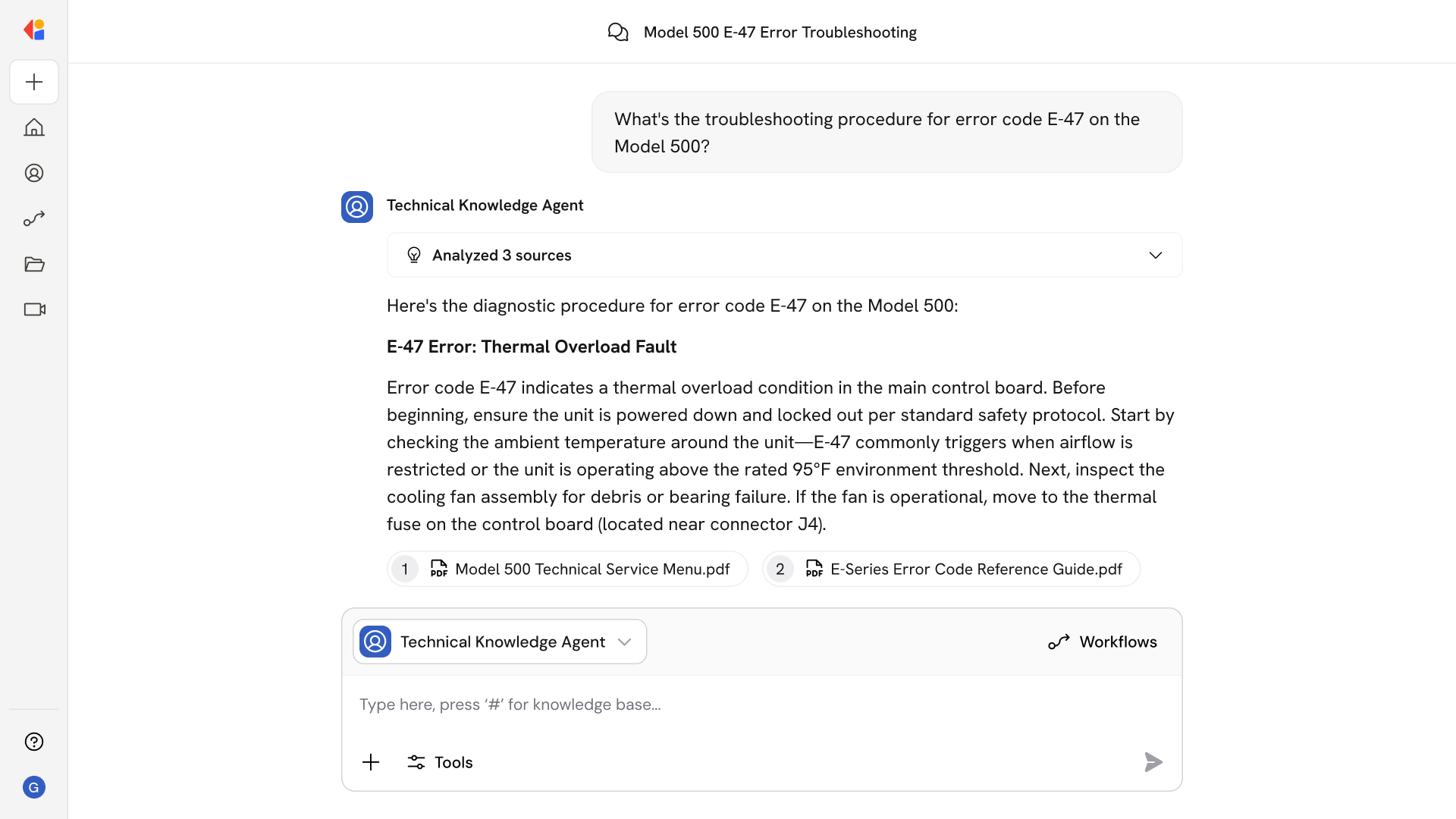
Step 1: Trigger on quality signal or scheduled review
The Workflow activates when a safety event is reported, a dashboard threshold is breached, a regulatory gap surfaces, or a scheduled audit cycle begins—pulling in relevant context automatically.
Step 2: Retrieve and synthesize evidence
Cassidy searches the Knowledge Base for applicable clinical guidelines, eCQM specifications, HEDIS codes, local SOPs, and prior PDSA logs—returning grounded answers with citations to specific document versions and sections.
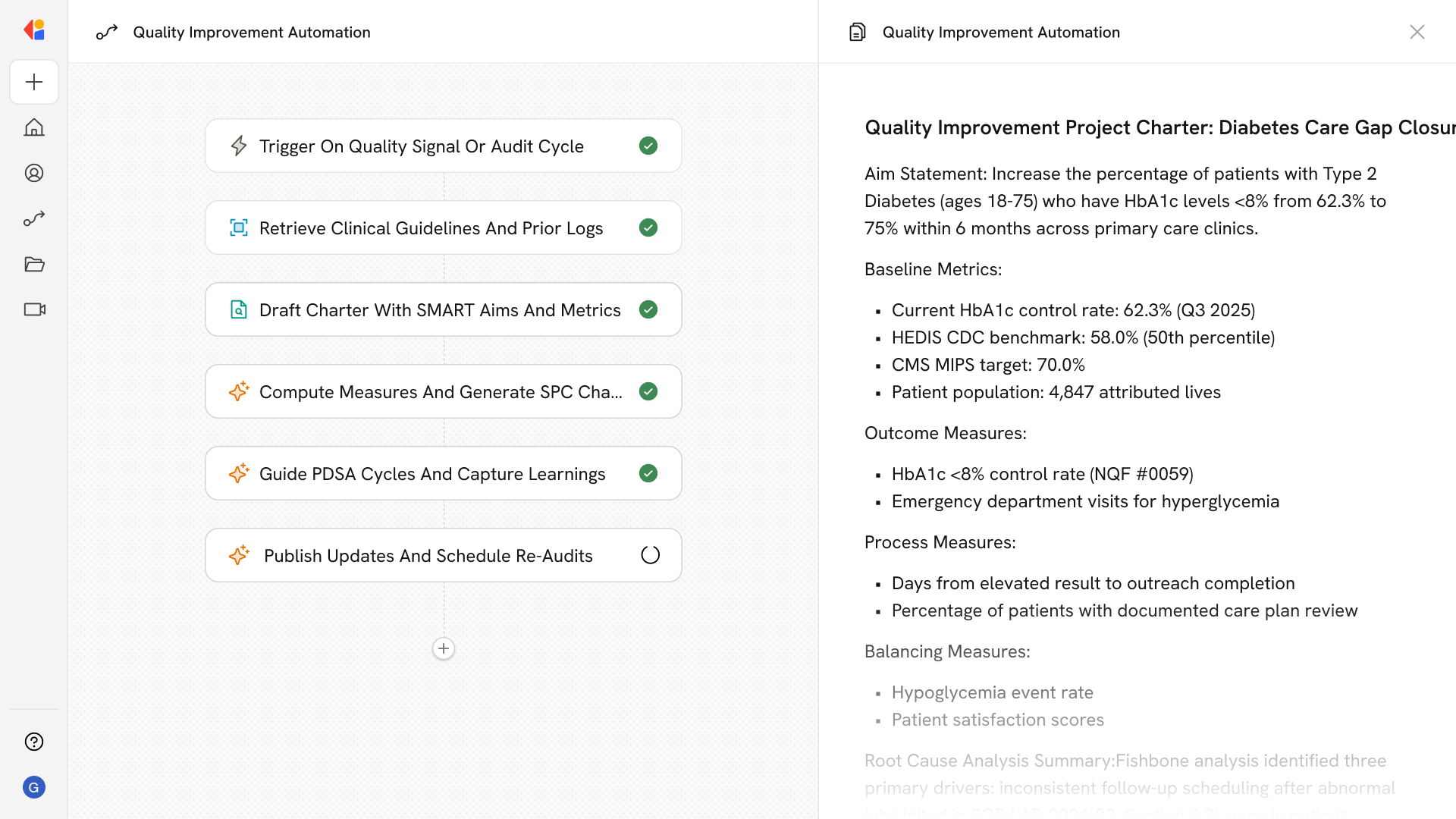
Step 3: Draft charter and measurement plan
The agent assembles a project charter with SMART aim statements, baseline metrics, and recommended outcome, process, and balancing measures aligned to CMS benchmarks and NQF stewardship.
Step 4: Compute measures and generate SPC charts
Cassidy connects to your EHR and EDW, applies measure logic with proper exclusions and risk adjustment, and produces run and control charts with equity stratification and benchmark comparisons.
Step 5: Guide PDSA cycles and capture learnings
The Workflow creates linked tests of change, collects cycle data, tracks predictions against results, and synthesizes lessons into KCS articles for reuse across the organization.
Step 6: Route for review with Human-in-the-Loop approval
All artifacts—RCA reports, driver diagrams, audit findings, and regulator-ready submissions—flow through configurable approval workflows with e-signatures and immutable audit trails.
Step 7: Publish and sustain
Approved content updates knowledge articles, refreshes standard work and order sets, and schedules re-audit reminders—maintaining an auditable record of data lineage, decisions, and version history for MIPS, HEDIS, and compliance reporting.
Implement it inside your company
- Hands-on onboarding and support
- Self-paced training for your team
- Dedicated implementation experts
- Ongoing use case discovery
- ROI tracking & analytics dashboards
- Proven playbooks to get started fast