AI FDA ISO Compliance Monitoring Agent
Automating FDA ISO Compliance Monitoring with AI
Centralized audit trail aggregation and review
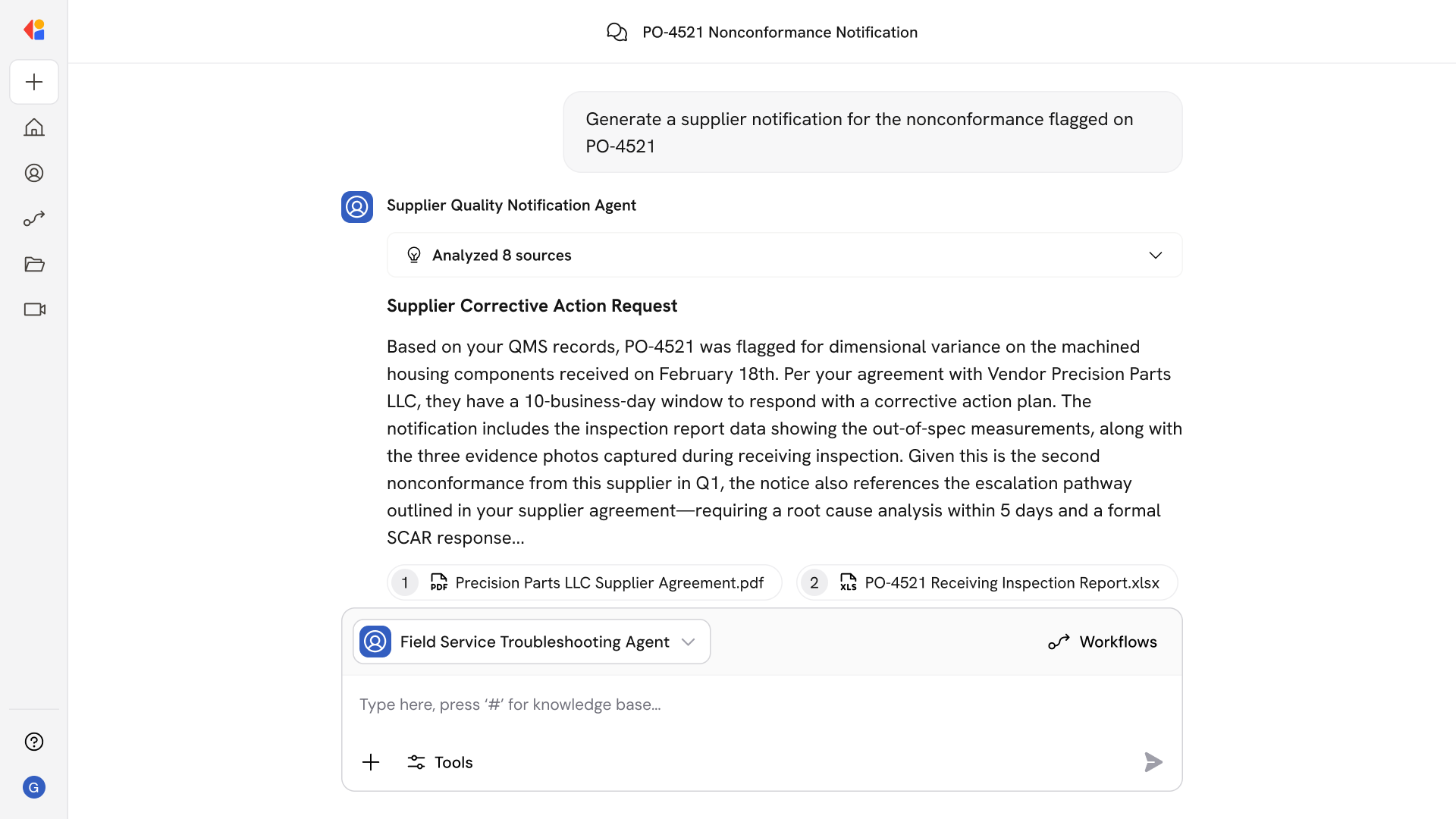
The agent pulls audit logs from MES, LIMS, eBR, and historians into a single immutable repository, enabling exception-based review that replaces manual, record-by-record reconciliation.
Continuous GxP data integrity monitoring
AI continuously evaluates ALCOA+ attributes across systems—flagging backdated entries, shared accounts, missing metadata, and segregation-of-duty conflicts before they become inspection findings.
Automated deviation and CAPA orchestration
When anomalies surface, the agent triages events, routes them for QA review, and guides investigations through root cause analysis, risk assessment, and effectiveness checks—with full traceability.
How Cassidy automates audit trail review and compliance workflows using AI
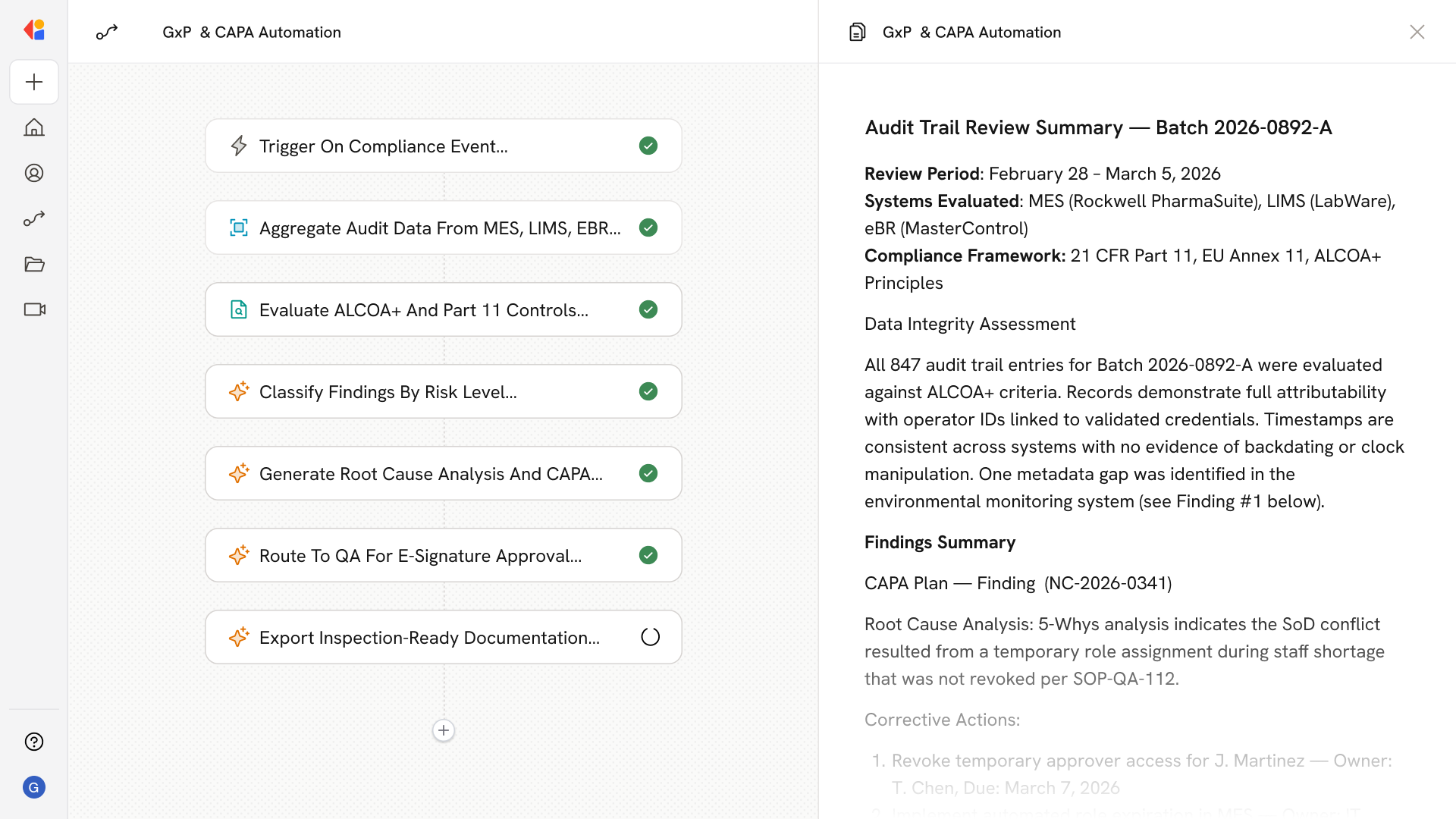
Step 1: Trigger on compliance event or scheduled review
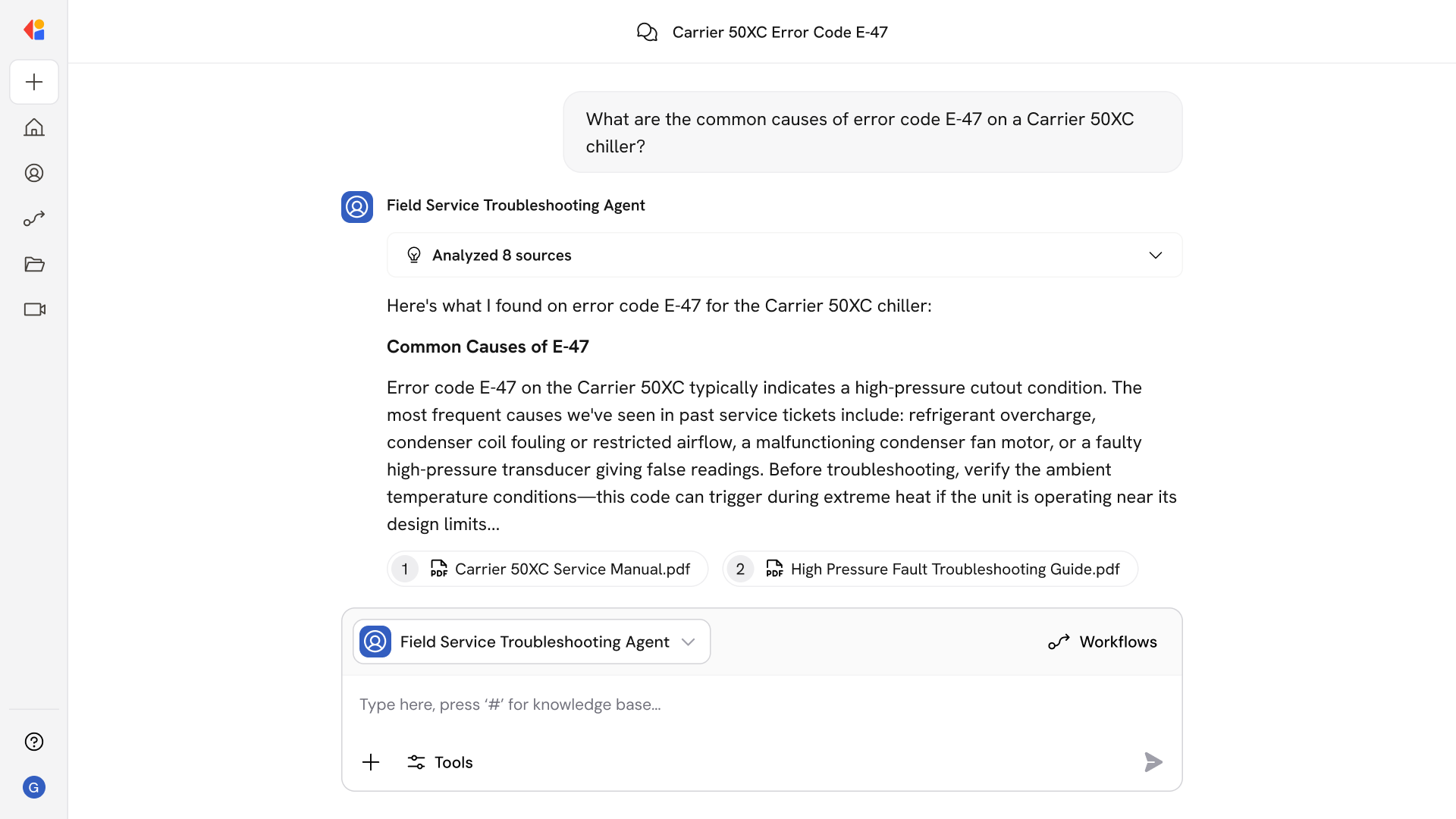
The Workflow activates when a new audit trail entry, deviation, or environmental excursion is detected—or on a scheduled cadence for periodic compliance reviews.
Step 2: Aggregate and normalize audit data
Cassidy connects to your MES, LIMS, eBR, historians, and document control systems to pull relevant records, normalizing data with full provenance (who, what, when, where) into a tamper-evident format.
Step 3: Evaluate ALCOA+ and Part 11/Annex 11 controls
The Agent checks each record against ALCOA+ data integrity principles and Part 11/Annex 11 requirements—detecting backdating, SoD conflicts, incomplete metadata, and unauthorized changes.
Step 4: Triage and classify findings
Cassidy categorizes detected issues as deviations, nonconformances, or incidents, assigns risk scores, and routes them to the appropriate QA reviewers with linked evidence.
Step 5: Orchestrate investigation and CAPA
For flagged events, the Workflow guides investigators through root cause analysis (5-Whys, Ishikawa), FMEA risk assessment, and CAPA plan generation—assigning owners, due dates, and effectiveness checks.
Step 6: Human-in-the-Loop approval
Quality reviewers receive structured cases with all supporting documentation, approve or request changes via e-signature, and attest to audit trail reviews per your SOPs.
Step 7: Generate inspection-ready evidence
Cassidy compiles audit trail exports, deviation/CAPA dossiers, validation artifacts, and training records into inspection-ready packages that preserve content and meaning.
Implement it inside your company
- Hands-on onboarding and support
- Self-paced training for your team
- Dedicated implementation experts
- Ongoing use case discovery
- ROI tracking & analytics dashboards
- Proven playbooks to get started fast